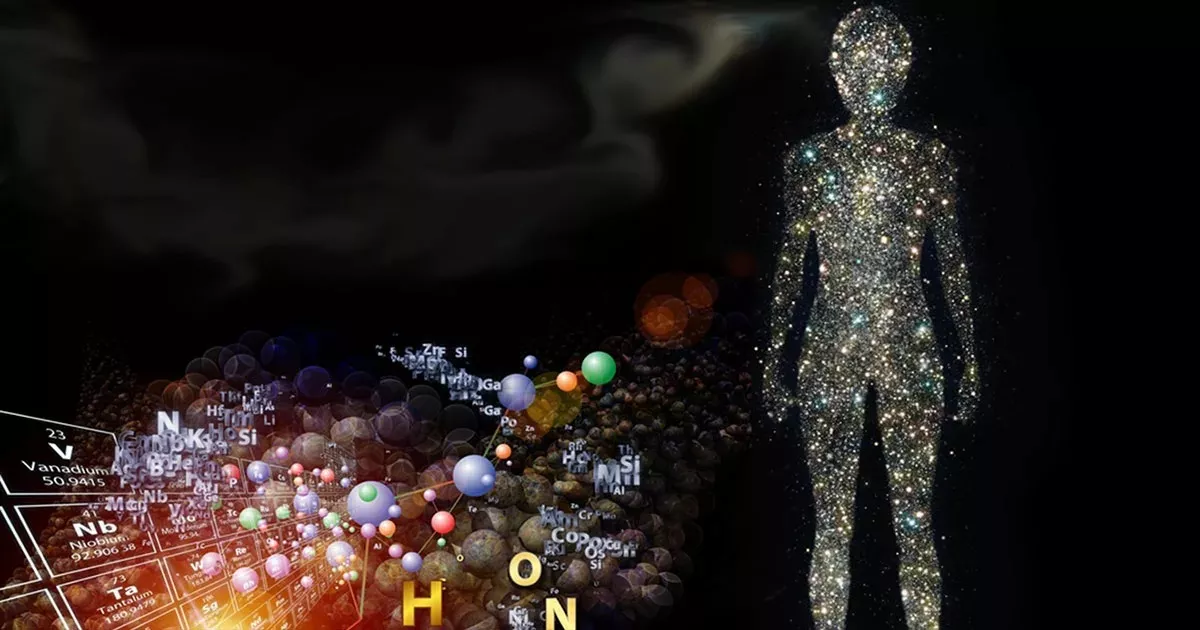

The Cosmos is within us; we're made of star stuff. We are a way for the cosmos to know itself.
~Carl Sagan
When Carl Sagan said, “We are made of star stuff,” he wasn’t just being poetic. He was speaking of a profound scientific truth. Everything we are, and everything we see, was forged in the heart of a star. To understand how this happens, we have to journey back to the beginning of the universe, when the only elements in existence were hydrogen and a tiny bit of helium. From these humble beginnings, a cosmic alchemy has unfolded over billions of years, transforming the simplest building blocks into the rich tapestry of elements that make up our bodies and the world around us.
In the early universe, vast clouds of hydrogen gas floated through space. Over time, these clouds started to clump together under the force of gravity, growing denser and hotter at their cores. When the pressure and temperature became immense enough, a remarkable thing happened—fusion began. This is the process where lighter elements combine to form heavier ones, releasing tremendous amounts of energy.
Imagine a pot of soup simmering on the stove. As the heat increases, the ingredients mix and merge, creating new flavors. Similarly, in the core of a star, hydrogen atoms are "cooked" together, forming helium. This process, called the proton-proton chain reaction, is the same one that powers our sun. As the star matures, it starts to create more complex elements, step by step.
Fusion is the process that drives stars and creates new elements. It all starts with hydrogen, the simplest and most abundant element in the universe. Picture two hydrogen atoms—essentially just protons—flying toward each other at unimaginable speeds. Despite being positively charged and naturally repelling each other, the extreme pressure and temperature inside a star force them to collide.
When they do, something extraordinary happens. One proton transforms into a neutron, creating a new particle called deuterium. As more protons join the mix, the deuterium atoms combine to form helium. But the process doesn’t stop there. As the star continues to "burn" its fuel, it starts creating heavier and heavier elements—each stage of fusion building on the last.
As the star exhausts its hydrogen supply, it begins to fuse helium in a process known as the triple-alpha process. This reaction involves three helium atoms coming together to form carbon, a fundamental element for life. In larger stars, fusion can continue beyond carbon, creating elements like oxygen, neon, and silicon. However, the process halts when it reaches iron.
Iron is a unique element in the fusion chain because, unlike lighter elements, its formation doesn’t release energy. Instead, it consumes it. Once a star starts producing iron, it has reached the end of its lifecycle. With no more energy being generated, gravity takes over, causing the star to collapse under its own weight.
When a massive star runs out of fuel and collapses, it explodes in a supernova—a dramatic and violent event that sends shockwaves through space. During these fleeting moments, the star becomes a cauldron of creation, forging elements heavier than iron, such as gold, uranium, and platinum.
This explosive finale scatters these newly formed elements across the universe, enriching the interstellar clouds with the building blocks for new stars, planets, and eventually, life. The next generation of stars and solar systems forms from this enriched material, creating even more diverse and complex elements.
After a supernova, what remains is often a beautiful, glowing cloud of gas and dust called a nebula. These are the nurseries of new stars. The elements scattered by the supernova mix with existing hydrogen and helium, creating the perfect environment for star formation. Gravity starts to pull the gas and dust together, and if there’s a nearby shockwave from another supernova or a strong gravitational force, the process of star birth begins anew.
It’s in these nebulae that new stars, planets, moons, and asteroids are formed. Every nebula is like a cosmic recipe book, with the right ingredients for making not just stars, but entire solar systems.
Take a moment to consider this: every atom in your body was once inside a star. The oxygen in your lungs, the calcium in your bones, and the iron in your blood were all created in the heart of a star that lived and died billions of years ago. When Carl Sagan said, “We are made of star stuff,” he meant it quite literally.
Our bodies are primarily composed of six elements: oxygen, hydrogen, nitrogen, carbon, calcium, and phosphorus. All of these elements were formed through stellar fusion and supernova explosions. They were scattered across space, eventually coming together to form planets, plants, animals, and people. This cosmic recycling process has been ongoing since the universe began.
The atoms that make up everything around us have been on an incredible journey, from the fiery furnaces of stars to the quiet coolness of Earth. It’s humbling to realize that the calcium in our bones might have come from a different star than the carbon in our DNA. Each breath you take, every beat of your heart, is a testament to the stars that lived and died to create the elements necessary for life.
In essence, we are part of the universe, not just in a poetic sense, but in a very real, tangible way. The universe isn’t something outside of us—it’s something we are intrinsically connected to. We are the cosmos made conscious, a way for the universe to look back upon itself and wonder.
Knowing how elements are made helps us understand our place in the universe. It shows us that we are not separate from the cosmos but a continuation of it. The atoms that once fueled stars now fuel our bodies, our minds, and our creativity. We are truly star stuff—connected to every star that has ever shone and every star that will ever shine.
So, the next time you look up at the night sky, remember: you’re not just gazing at distant lights. You’re looking at your origins. You’re looking at where you came from and where one day, long in the future, your atoms may return.